Has A Definite Volume And Shape
listenit
Apr 02, 2025 · 5 min read
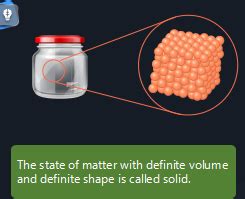
Table of Contents
Has a Definite Volume and Shape: Exploring the Properties of Solids
The phrase "has a definite volume and shape" is a key characteristic used to define a state of matter: the solid state. Understanding this property is fundamental to comprehending the behavior of materials in our world, from the skyscrapers that pierce the sky to the intricate structures within our own bodies. This article delves deep into the reasons behind this defining characteristic, exploring the atomic structure, intermolecular forces, and macroscopic properties that contribute to the rigidity and stability of solids.
The Atomic Basis of Definite Volume and Shape
At the heart of a solid's definite volume and shape lies its atomic structure. Unlike gases, whose atoms or molecules are widely dispersed and move freely, and liquids, where particles maintain close proximity but still exhibit some mobility, the constituent particles of a solid are tightly packed together in a highly ordered arrangement. This arrangement, often referred to as a crystal lattice or a crystalline structure, is a three-dimensional, repeating pattern of atoms, ions, or molecules.
Crystalline Solids: The Ordered Arrangement
Many solids are crystalline, meaning their constituent particles are arranged in a highly ordered, repeating pattern. This order extends over a large scale, creating a long-range order. Examples include table salt (sodium chloride), diamonds (carbon), and quartz (silicon dioxide). The specific arrangement depends on the types of atoms involved and the forces between them. The strength and type of these forces dictate the properties of the solid.
-
Ionic Solids: These solids are formed by electrostatic attraction between oppositely charged ions. For example, in sodium chloride (NaCl), the positively charged sodium ions (Na⁺) and negatively charged chloride ions (Cl⁻) are arranged in a regular cubic lattice, maximizing electrostatic attraction and minimizing repulsion. This strong attraction contributes to the hardness and high melting points typical of ionic solids. Their definite volume and shape are directly attributable to the strong forces holding the ions in place.
-
Covalent Solids: In covalent solids, atoms are linked by strong covalent bonds, creating a network of interconnected atoms. Diamond, a classic example, consists of a three-dimensional network of carbon atoms, each bonded to four others via strong covalent bonds. This strong bonding accounts for the exceptional hardness and high melting point of diamond. The fixed positions of atoms in the rigid network contribute directly to its definite volume and shape.
-
Metallic Solids: Metallic solids consist of a lattice of metal cations surrounded by a "sea" of delocalized electrons. These electrons are not bound to specific atoms, but rather move freely throughout the metal lattice. This arrangement explains the high electrical and thermal conductivity of metals. The metallic bonds, while not as directional as covalent bonds, are strong enough to maintain the definite volume and shape of the solid.
Amorphous Solids: The Lack of Long-Range Order
While many solids are crystalline, some exhibit amorphous structures. In amorphous solids, the atoms or molecules are arranged randomly, lacking the long-range order found in crystalline materials. Examples include glass, rubber, and many plastics. While amorphous solids have a definite volume, their lack of long-range order results in them not possessing a definite shape. They can be molded or deformed more easily than crystalline solids. However, their definite volume stems from the strong intermolecular forces that prevent the particles from spreading out significantly.
Intermolecular Forces and their Role
The intermolecular forces between the atoms or molecules in a solid are crucial in determining its volume and shape. These forces are much weaker than the ionic or covalent bonds within molecules, but their collective effect is significant in determining the macroscopic properties of the solid.
Strong Intermolecular Forces: Maintaining Structure
Stronger intermolecular forces lead to solids with higher melting points, greater hardness, and more resistance to deformation. The particles are held more tightly in place, resulting in a more rigid structure and a well-defined volume and shape. Examples include hydrogen bonding in ice, which gives it a relatively high melting point for its molecular weight.
Weaker Intermolecular Forces: More Flexibility
Solids with weaker intermolecular forces tend to be softer and have lower melting points. The particles are not held as rigidly, allowing for greater flexibility and potentially less well-defined shapes. However, even with weaker forces, the particles still maintain close proximity, resulting in a definite volume.
Macroscopic Properties Reflecting the Microscopic Structure
The definite volume and shape of solids are reflected in various macroscopic properties:
-
Rigidity and Hardness: Solids resist deformation due to the strong intermolecular forces and ordered atomic arrangements. They maintain their shape unless subjected to significant external forces.
-
Incompressibility: Solids are nearly incompressible because their constituent particles are already closely packed together. Applying pressure only slightly reduces the interparticle distances.
-
Fixed Shape: The fixed positions of the particles in the solid's lattice determine its fixed shape. Crystalline solids possess a highly ordered, repeating pattern, leading to a well-defined geometric shape.
Exceptions and Special Cases
While the vast majority of solids exhibit definite volume and shape, some exceptions and special cases warrant consideration.
-
Polymorphism: Some substances can exist in more than one crystalline form, exhibiting different arrangements of atoms and thus different properties. This phenomenon, known as polymorphism, affects the material's shape and other physical characteristics.
-
Allotropes: These are different structural modifications of the same element. Carbon, for example, exists as diamond (highly ordered, very hard) and graphite (layered structure, soft). While both are solids, their vastly different structures lead to stark differences in their macroscopic properties, including shape and hardness.
Conclusion: The Interplay of Forces and Structure
The definite volume and shape of solids are a consequence of the strong intermolecular forces and highly ordered atomic arrangement characteristic of the solid state. The specific type of bonding and the strength of intermolecular interactions dictate the hardness, rigidity, and overall macroscopic properties of the solid. While exceptions and special cases exist, the fundamental principle remains consistent: the tightly packed and ordered nature of the constituent particles is the cornerstone of the solid's defining characteristics. Understanding this relationship between microscopic structure and macroscopic properties is crucial in materials science, engineering, and many other fields. From designing new materials with specific properties to understanding geological formations, the principles discussed here are fundamental to numerous scientific disciplines. Further research into the intricacies of solid-state physics and materials science will undoubtedly reveal even more fascinating aspects of this fundamental state of matter.
Latest Posts
Latest Posts
-
Whats The Square Root Of 500
Apr 03, 2025
-
How Many Feet Is 1 2 Miles
Apr 03, 2025
-
How Many Pounds In One Pint
Apr 03, 2025
-
Compute And Interpret The Mean Of The Random Variable X
Apr 03, 2025
-
Where Does Carbon Dioxide Enter A Plant
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Has A Definite Volume And Shape . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
