Are Hydrogen Bonds Stronger Than Ionic Bonds
listenit
Apr 04, 2025 · 5 min read
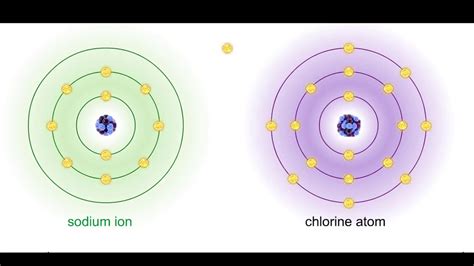
Table of Contents
Are Hydrogen Bonds Stronger Than Ionic Bonds? A Deep Dive into Intermolecular Forces
The question of whether hydrogen bonds are stronger than ionic bonds is a common point of confusion in chemistry. The short answer is no, ionic bonds are generally much stronger than hydrogen bonds. However, the full picture is far more nuanced and depends heavily on the specific context. This article will delve into the nature of both ionic and hydrogen bonds, comparing their strengths, providing examples, and clarifying misconceptions.
Understanding Ionic Bonds: The Electrostatic Attraction
Ionic bonds are formed through the electrostatic attraction between oppositely charged ions. This attraction arises from the complete transfer of one or more electrons from a metal atom to a non-metal atom. The metal atom loses electrons, becoming a positively charged cation, while the non-metal atom gains electrons, becoming a negatively charged anion. The strong Coulombic force between these ions holds them together in a crystal lattice structure.
Key Characteristics of Ionic Bonds:
- High Strength: The electrostatic forces involved are substantial, leading to high melting and boiling points.
- Crystalline Structure: Ionic compounds typically form well-ordered crystal lattices.
- Solubility in Polar Solvents: Ionic compounds often dissolve readily in polar solvents like water, due to the interaction between the ions and the polar solvent molecules.
- Electrical Conductivity: Ionic compounds conduct electricity when molten or dissolved in a solution, as the ions become mobile.
- Hardness and Brittleness: Ionic crystals are typically hard but brittle, as the displacement of ions can lead to repulsive forces.
Examples of Ionic Bonds: Sodium chloride (NaCl), magnesium oxide (MgO), potassium iodide (KI). These compounds are characterized by significant differences in electronegativity between the constituent atoms.
Understanding Hydrogen Bonds: A Special Type of Dipole-Dipole Interaction
Hydrogen bonds are a special type of dipole-dipole interaction. They occur when a hydrogen atom bonded to a highly electronegative atom (typically fluorine, oxygen, or nitrogen) is attracted to another electronegative atom in a different molecule (or a different part of the same molecule). This electronegative atom pulls the shared electrons away from the hydrogen atom, creating a partially positive charge (δ+) on the hydrogen and a partially negative charge (δ-) on the electronegative atom. The attraction between these partially charged atoms forms the hydrogen bond.
Key Characteristics of Hydrogen Bonds:
- Relatively Weak Strength: Compared to ionic or covalent bonds, hydrogen bonds are relatively weak.
- Directional Nature: Hydrogen bonds are directional, meaning they are strongest when the atoms are aligned in a straight line.
- Influence on Physical Properties: Hydrogen bonds significantly affect the physical properties of molecules, influencing melting and boiling points, solubility, and viscosity.
- Importance in Biological Systems: Hydrogen bonds play a crucial role in the structure and function of biological molecules like proteins and DNA.
- Cooperative Effects: The strength of a hydrogen bond can be influenced by the presence of other hydrogen bonds nearby; this is known as cooperative effect.
Examples of Hydrogen Bonds: Water (H₂O), ammonia (NH₃), and DNA base pairing. The high boiling point of water, for instance, is a direct result of strong hydrogen bonding between water molecules.
Comparing the Strengths: Bond Energy and Enthalpy
The strength of a bond is often quantified by its bond energy or bond enthalpy. Bond energy represents the energy required to break one mole of a particular bond in the gaseous phase. Ionic bonds generally have much higher bond energies than hydrogen bonds.
For example, the bond energy of NaCl is approximately 787 kJ/mol, while the hydrogen bond energy in water is around 20-25 kJ/mol. This significant difference underscores the substantially greater strength of ionic bonds.
It is important to remember that hydrogen bond strength can vary depending on several factors, including:
- The electronegativity of the atoms involved: Higher electronegativity differences lead to stronger hydrogen bonds.
- The geometry of the molecules: Linear arrangements maximize hydrogen bond strength.
- The surrounding environment: The presence of other molecules or ions can affect the hydrogen bond strength.
Misconceptions and Clarifications
One common misconception is that because hydrogen bonds are crucial in biological systems, they must be incredibly strong. While hydrogen bonds are essential for the structure and function of biological macromolecules like proteins and DNA, they are still relatively weak compared to ionic bonds. The collective effect of numerous hydrogen bonds contributes to the overall stability of these biological structures.
Another misconception arises from the high boiling point of water. While the high boiling point is indeed due to hydrogen bonding, it does not imply that hydrogen bonds are stronger than ionic bonds. The high boiling point is a result of the cumulative effect of numerous hydrogen bonds between water molecules, not the intrinsic strength of a single hydrogen bond.
Conclusion: Context is Key
In summary, ionic bonds are significantly stronger than hydrogen bonds. While hydrogen bonds play vital roles in many biological and chemical processes, their strength pales in comparison to the robust electrostatic attraction of ionic bonds. The key takeaway is that the relative strength of different intermolecular forces must be considered within their specific context. While ionic bonds dominate in terms of individual bond strength, the collective effect of numerous weaker hydrogen bonds can have profound impacts on macroscopic properties and molecular interactions. Understanding these differences is fundamental to grasping the behavior of various chemical systems.
Further Exploration: Beyond the Basics
Beyond the fundamental comparison of ionic and hydrogen bonds, we can further explore:
- Covalent Bonds: How do the strengths of ionic and hydrogen bonds compare to covalent bonds? Covalent bonds, involving the sharing of electrons, are stronger than hydrogen bonds but generally weaker than ionic bonds, although there are many exceptions.
- Van der Waals forces: How do weaker intermolecular forces like London Dispersion Forces and dipole-dipole interactions fit into this picture? These forces are generally much weaker than hydrogen bonds and ionic bonds.
- Factors influencing bond strength: A deeper dive into the factors influencing the strength of both ionic and hydrogen bonds, including factors such as charge density, size of the ions, and solvent effects, would provide a more thorough understanding.
- Applications in Materials Science: Understanding the relative strengths of these bonds is critical in materials science, affecting material properties such as melting point, solubility, and mechanical strength.
This in-depth exploration provides a more comprehensive understanding of the fascinating world of intermolecular forces and their influence on the physical and chemical properties of matter. The seemingly simple question of "are hydrogen bonds stronger than ionic bonds" unveils a complex interplay of forces governing the behavior of the universe around us.
Latest Posts
Latest Posts
-
How Many Moles Are 1 20 X10 25 Atoms Of Phosphorus
Apr 04, 2025
-
Which Element Is The Least Reactive
Apr 04, 2025
-
What Is The Energy Transformation That Occurs During Photosynthesis
Apr 04, 2025
-
Number Of Valence Electrons Of Calcium
Apr 04, 2025
-
How Many Neutrons Are In Sulfur
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Are Hydrogen Bonds Stronger Than Ionic Bonds . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
