All Atoms Of A Given Element Have The Same
listenit
Apr 01, 2025 · 6 min read
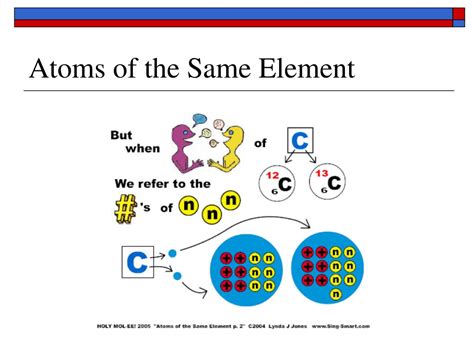
Table of Contents
- All Atoms Of A Given Element Have The Same
- Table of Contents
- All Atoms of a Given Element Have the Same: Delving into Atomic Structure and Isotopes
- Understanding the Building Blocks: Protons, Neutrons, and Electrons
- The Significance of Atomic Number
- Isotopes: Variations on a Theme
- Impact of Isotopes on Atomic Mass
- Isotopic Properties: Similarities and Differences
- Radioactivity and Isotopes
- Beyond the Basics: A Deeper Look into Atomic Structure
- The Periodic Table: Organizing the Elements
- Applications and Implications
- Conclusion: Refining the Initial Statement
- Latest Posts
- Latest Posts
- Related Post
All Atoms of a Given Element Have the Same: Delving into Atomic Structure and Isotopes
The statement "all atoms of a given element have the same" is a simplification, a foundational concept in chemistry that requires nuanced understanding. While fundamentally true in terms of atomic number, the reality is more intricate when considering isotopes and their subtle variations in mass. This article will delve into the intricacies of atomic structure, exploring the aspects that remain consistent and the variations that exist within a single element.
Understanding the Building Blocks: Protons, Neutrons, and Electrons
Atoms, the fundamental units of matter, are composed of three subatomic particles:
- Protons: Positively charged particles located in the atom's nucleus.
- Neutrons: Neutral particles (no charge) also residing in the nucleus.
- Electrons: Negatively charged particles orbiting the nucleus in electron shells or energy levels.
The defining characteristic of an element lies in its atomic number, which represents the number of protons in its nucleus. This number is unique to each element and is crucial for its chemical identity and properties. All atoms of a given element possess the same atomic number. This is the core truth behind the statement in the title. For example, all hydrogen atoms have one proton, all carbon atoms have six protons, and all oxygen atoms have eight protons. This consistent proton number dictates the element's position on the periodic table and largely determines its chemical behavior.
The Significance of Atomic Number
The atomic number dictates the number of electrons an atom possesses in its neutral state. Since atoms strive for stability (a full outermost electron shell), this number heavily influences how an element interacts with other elements, forming chemical bonds and compounds. The consistent atomic number ensures predictable and repeatable chemical reactions. Without this consistent proton count, the very foundation of chemistry would collapse into chaos.
Isotopes: Variations on a Theme
While all atoms of a given element share the same atomic number (proton count), they can differ in their number of neutrons. These variations are called isotopes. Isotopes of the same element have the same number of protons but different numbers of neutrons. This difference in neutron number leads to a variation in the atom's mass number (protons + neutrons).
For example, carbon-12 (¹²C), the most common isotope of carbon, has six protons and six neutrons. Carbon-13 (¹³C) also has six protons but possesses seven neutrons, resulting in a higher mass. Similarly, Carbon-14 (¹⁴C), a radioactive isotope used in carbon dating, has six protons and eight neutrons.
Impact of Isotopes on Atomic Mass
The differences in neutron numbers influence the mass of the atom, affecting its overall weight. The standard atomic weight listed on the periodic table is a weighted average of the masses of all naturally occurring isotopes of an element, considering their relative abundances. This explains why atomic weights are not always whole numbers. The abundance of each isotope varies for different elements.
Isotopic Properties: Similarities and Differences
Despite their mass differences, isotopes of the same element largely exhibit similar chemical properties. This is because the number of protons and electrons, which determine chemical reactivity, remains constant. The differences primarily manifest in physical properties such as mass and sometimes radioactivity (in the case of radioactive isotopes).
Radioactivity and Isotopes
Some isotopes are unstable, meaning their nuclei tend to decay over time, emitting radiation in the process. This phenomenon is known as radioactivity. Radioactive isotopes are used in various applications, including medical imaging, cancer treatment, and archaeological dating. The rate of decay is characteristic of each radioactive isotope and is described by its half-life.
Radioactive decay changes the number of protons in the nucleus, thus transforming the atom into an atom of a different element. This transmutation is a fundamental process in nuclear chemistry and physics.
Beyond the Basics: A Deeper Look into Atomic Structure
The simple model of electrons orbiting the nucleus like planets around the sun is an oversimplification. Quantum mechanics provides a more accurate, albeit more complex, picture of atomic structure. Electrons don't follow fixed orbits but exist in atomic orbitals, regions of space where the probability of finding an electron is high. These orbitals have different shapes and energy levels, which influence the atom's chemical behavior.
The electron configuration, the arrangement of electrons in these orbitals, determines the chemical properties of an atom. The outermost electrons, called valence electrons, are particularly important as they participate in chemical bonding. All atoms of the same element have the same number of protons and, in their neutral state, the same number of electrons, leading to the same electron configuration and hence similar chemical behavior.
The Periodic Table: Organizing the Elements
The periodic table is a systematic arrangement of elements based on their atomic number and electronic structure. Elements with similar electronic configurations and, consequently, similar chemical properties, are grouped together in columns (groups or families). This arrangement reflects the periodic trends observed in the properties of elements, such as electronegativity, ionization energy, and atomic radius.
The periodic table's structure directly relates to the consistent proton number defining each element. Without this consistency, the table wouldn't be able to organize elements based on predictable chemical behaviors. The table is a testament to the underlying principle that all atoms of a given element possess the same number of protons.
Applications and Implications
The principle that all atoms of a given element have the same atomic number is fundamental to many scientific and technological advancements. Some key applications include:
- Spectroscopy: The unique spectral lines emitted or absorbed by elements are due to the specific arrangement of electrons, directly tied to the atomic number. This is used for elemental analysis in various fields.
- Nuclear Chemistry: Understanding isotopes and their radioactive decay is crucial for applications in medicine, energy production, and environmental monitoring.
- Material Science: The properties of materials are heavily influenced by the arrangement and interactions of atoms. The knowledge of atomic structure and bonding allows for the design and synthesis of new materials with specific properties.
- Chemical Synthesis: Chemical reactions rely on the predictable behavior of atoms based on their electronic configuration. Understanding the consistent atomic number allows for the precise synthesis of desired compounds.
Conclusion: Refining the Initial Statement
The statement "all atoms of a given element have the same" needs refinement. While it's accurate in stating that all atoms of a given element possess the same atomic number (number of protons), this doesn't encompass the variations that arise from different numbers of neutrons, leading to isotopes. Isotopes of the same element exhibit subtle differences in mass and sometimes radioactivity, but their chemical properties remain largely consistent due to the identical number of protons and electrons in their neutral state. The consistent atomic number remains the cornerstone of chemistry, providing the foundation for the periodic table and enabling a vast array of scientific and technological applications. The existence of isotopes adds complexity, enriching our understanding of the intricacies of matter at the atomic level. This nuanced understanding, encompassing both the constancy of atomic number and the variability of isotopes, is crucial for a comprehensive grasp of the behavior and properties of elements.
Latest Posts
Latest Posts
-
Which Function Has A Negative Discriminant Value
Apr 04, 2025
-
A Number Y Is No More Than
Apr 04, 2025
-
Why Are Hydrogen Bonds Important To Life
Apr 04, 2025
-
How Much Is 9 Ounces In Cups
Apr 04, 2025
-
Describe The Function Of The Lens
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about All Atoms Of A Given Element Have The Same . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
