6 Protons 6 Neutrons 6 Electrons Total Charge
listenit
Apr 06, 2025 · 6 min read
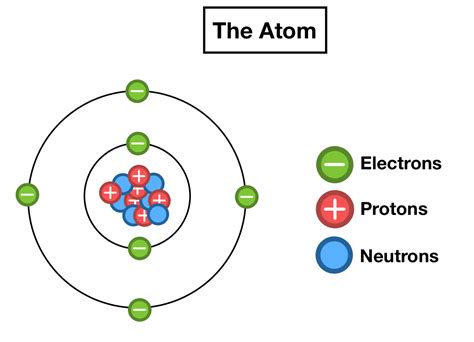
Table of Contents
6 Protons, 6 Neutrons, 6 Electrons: Exploring the Atom of Carbon-12
The statement "6 protons, 6 neutrons, 6 electrons" immediately points to a specific atom: carbon-12 (¹²C). Understanding this atom's composition and its implications is crucial to grasping fundamental concepts in chemistry and physics. This article delves deep into the structure of carbon-12, exploring its properties, significance, and the implications of its specific proton, neutron, and electron count.
Understanding Subatomic Particles: Protons, Neutrons, and Electrons
Before we delve into the specifics of carbon-12, let's refresh our understanding of the three fundamental subatomic particles:
Protons: The Positive Charge Carriers
Protons are positively charged particles found within the atom's nucleus. The number of protons defines the element; an atom with six protons is always a carbon atom. The proton's mass is approximately 1 atomic mass unit (amu). The positive charge of the proton is crucial in determining the atom's overall charge and its interactions with other atoms.
Neutrons: The Neutral Nucleus Residents
Neutrons, as their name suggests, carry no electrical charge. They reside alongside protons in the atom's nucleus. A neutron's mass is slightly larger than a proton's, also approximately 1 amu. Neutrons play a vital role in stabilizing the nucleus, especially in heavier atoms. The number of neutrons in an atom can vary, leading to different isotopes of the same element.
Electrons: The Orbiting Negatives
Electrons are negatively charged particles that orbit the nucleus in specific energy levels or shells. They are significantly lighter than protons and neutrons, with a mass approximately 1/1836 that of a proton. The number of electrons in a neutral atom is equal to the number of protons. These electrons determine the atom's chemical properties and its ability to form bonds with other atoms.
Carbon-12: The Isotope Standard
Carbon-12, with its six protons, six neutrons, and six electrons, holds a unique position in the world of chemistry and physics. Its significance stems from its role as the standard for atomic mass units.
Atomic Mass Units (amu): Defining the Standard
The atomic mass unit (amu) is defined as 1/12 the mass of a single carbon-12 atom. This means that the mass of a carbon-12 atom is exactly 12 amu. All other atomic masses are relative to this standard. The choice of carbon-12 as the standard is arbitrary but convenient, given its abundance and relatively simple structure.
Isotopes: Variations on a Theme
Isotopes are atoms of the same element that have the same number of protons but a different number of neutrons. Carbon has several isotopes, the most common being carbon-12 (¹²C), carbon-13 (¹³C), and carbon-14 (¹⁴C). While ¹²C has six neutrons, ¹³C has seven and ¹⁴C has eight. These variations in neutron numbers affect the atom's mass but not its chemical properties significantly. However, the differing nuclear stability leads to different applications, for example, carbon-14 dating.
The Significance of the 6:6:6 Ratio in Carbon-12
The specific 6:6:6 ratio of protons, neutrons, and electrons in carbon-12 contributes to its unique properties:
-
Nuclear Stability: The even number of protons and neutrons contributes to the nucleus's relative stability. This stability is important in determining the element's half-life and its interactions in nuclear reactions. A stable nucleus doesn't readily undergo radioactive decay.
-
Chemical Reactivity: The six electrons in carbon-12 are arranged in two electron shells: two electrons in the innermost shell (K shell) and four electrons in the outermost shell (L shell). This configuration results in carbon's ability to form four covalent bonds, making it a fundamental building block of organic molecules. This tetravalency is responsible for the immense diversity of organic compounds.
-
Abundance in Nature: Carbon-12 is the most abundant isotope of carbon, comprising approximately 98.9% of all carbon atoms found in nature. Its abundance contributes to its importance in various biological and geological processes.
Applications and Importance of Carbon-12
Carbon-12's unique properties make it essential in various fields:
-
Organic Chemistry: As the backbone of all organic molecules, carbon-12 plays a crucial role in the study of life and the vast array of organic compounds. From simple hydrocarbons to complex biomolecules like proteins and DNA, carbon forms the structural basis of these vital molecules.
-
Nuclear Physics: Carbon-12 serves as a standard in nuclear physics research. Its well-understood structure and behavior make it a reference point for studying nuclear reactions and interactions. Scientists use carbon-12 in experiments to study nuclear forces and properties.
-
Mass Spectrometry: In mass spectrometry, carbon-12 is utilized as a reference for determining the masses of other molecules. The precise mass of carbon-12 helps calibrate instruments and ensure accurate measurements.
-
Radiocarbon Dating: While carbon-12 is stable, the radioactive isotope carbon-14 is used in radiocarbon dating to determine the age of organic materials. By comparing the ratio of ¹⁴C to ¹²C, scientists can estimate how long ago the organism lived.
Implications of Variations in Proton, Neutron, and Electron Numbers
Any alteration to the 6:6:6 ratio in carbon-12 results in significant changes:
-
Changing the Number of Protons: Changing the number of protons alters the element itself. Adding or subtracting a proton transforms the atom into a different element entirely (e.g., 5 protons would be boron, 7 protons would be nitrogen).
-
Changing the Number of Neutrons: Changing the number of neutrons creates an isotope of carbon. Isotopes have slightly different masses but generally share the same chemical properties. However, nuclear stability can be significantly affected. For instance, carbon-14 is radioactive, while carbon-12 and carbon-13 are stable.
-
Changing the Number of Electrons: Changing the number of electrons results in an ion—a charged species. If you remove one electron, you create a positively charged carbon ion (cation). Adding an electron creates a negatively charged ion (anion). Ions have different chemical properties compared to neutral atoms.
Conclusion: The Fundamental Importance of Carbon-12
The seemingly simple statement "6 protons, 6 neutrons, 6 electrons" encapsulates the essence of carbon-12, a fundamental atom with profound implications across various scientific disciplines. Its stable nucleus, tetravalent nature, and abundance in nature make it a cornerstone of life and crucial for understanding the building blocks of matter. The precise definition of atomic mass units based on carbon-12 highlights its importance as a fundamental standard in scientific measurement. Understanding the role of protons, neutrons, and electrons in determining an atom's properties is key to comprehending the behavior of matter at the atomic level. Carbon-12, with its 6:6:6 composition, stands as a testament to the elegant simplicity and profound significance of fundamental atomic structure.
Latest Posts
Latest Posts
-
93 Is What Percent Of 186
Apr 06, 2025
-
How To Find All Zeros Of A Polynomial
Apr 06, 2025
-
Domain And Range For X 3
Apr 06, 2025
-
When Water Freezes Ice Floats Why
Apr 06, 2025
-
Write The Electron Configuration For A Neutral Atom Of Tin
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about 6 Protons 6 Neutrons 6 Electrons Total Charge . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
